Hormone replacement therapy prescriptions for women aged 50–65 have increased 86% since 2021. The FDA removed boxed warnings on HRT products last year, citing evolving evidence on safety and newer delivery methods. Menopause awareness has gone mainstream, driven by high-profile advocacy, media coverage, and a generation of women demanding better care. By every measure, 2026 should be the best year in decades for women’s access to hormone therapy.
Instead, prescribers are fielding calls from patients who can’t fill their estradiol patch prescriptions.
The estrogen transdermal patch shortage—driven by a convergence of surging demand, limited manufacturers, tariff pressures on overseas production, and the ripple effects of Bayer’s 2023 discontinuation of its Climara patch—has created a real-time access crisis for women on stable HRT regimens. Major pharmacy chains are reporting delays. Patients are scrambling between pharmacies. Prescribers are rewriting prescriptions for alternative formulations mid-cycle.
This moment is forcing a broader conversation about women’s HRT access—one that every prescriber should be part of. Here’s what’s happening, what your options are, and where compounding pharmacy fits into the picture.
What’s Driving the Estrogen Patch Shortage
The shortage isn’t a single-cause problem. It’s the predictable result of demand growth colliding with supply fragility.
Demand surge. The FDA’s removal of boxed warnings on HRT products was a watershed moment. For two decades after the 2002 Women’s Health Initiative, prescribers were cautious about HRT—and many avoided prescribing it altogether. The warning removal, combined with updated clinical guidelines and growing patient demand, triggered a prescribing surge that the supply chain was not built to absorb. HRT prescriptions for women are up 86% since 2021, according to Epic Research data, and the growth shows no sign of slowing.
Manufacturer consolidation. After Bayer discontinued its Climara estradiol patch in late 2023, the U.S. transdermal estrogen market consolidated around just three primary manufacturers: Sandoz, Viatris (formerly Mylan), and Amneal. When demand spikes against a concentrated supply base, shortages propagate faster and resolve more slowly. There simply aren’t enough production lines to flex capacity quickly.
Tariff and supply chain pressure. Tariffs on overseas pharmaceutical manufacturing have added cost and complexity to the transdermal patch supply chain, with several estradiol patch components sourced internationally. These pressures compound (no pun intended) the demand-supply imbalance and make the shortage timeline less predictable.
The clinical preference for transdermal delivery. Transdermal estradiol carries a lower risk of blood clots compared to oral formulations because it bypasses first-pass hepatic metabolism. This safety advantage has made patches the preferred delivery method for many prescribers—which means the shortage is hitting the formulation that clinicians most want to prescribe.
The Menopause Care Gap: A Shortage on Top of a Shortage
The estrogen patch shortage is making headlines, but it’s layered on top of a deeper structural problem: the menopause care gap itself.
The prescriber shortage is real. Data from over 5,400 women treated between 2016 and 2023 shows that only 17% of women seeking menopause-related care received prescription treatment for their symptoms—with dramatic variability across provider specialties. Many primary care physicians, internists, and family medicine providers received minimal menopause training during residency. The result is that millions of symptomatic women either don’t receive treatment, receive suboptimal treatment, or face long wait times to see a menopause-trained specialist.
Access is unevenly distributed. Women in rural areas, women without specialist access, and women whose prescribers are not comfortable initiating HRT face disproportionate barriers. When you add a supply shortage on top of a prescriber shortage, the access gap widens further—and the patients who were already underserved bear the greatest burden.
The conversation is changing. The positive trend in all of this is awareness. Menopause advocacy organizations, celebrity voices, and legislative action (including state-level menopause workplace accommodation bills) have pushed women’s midlife health into the mainstream. More women are asking for HRT. More prescribers are willing to prescribe it. The infrastructure to deliver it—supply chain, trained providers, accessible pharmacy options—is what needs to catch up.
Where Compounded HRT Fits—and Where It Doesn’t
Compounded hormone therapy is not a blanket replacement for FDA-approved products. That distinction matters, and prescribers should be clear-eyed about it.
FDA-approved HRT products should remain the first-line option when they are available and meet the patient’s clinical needs. Dozens of FDA-approved estrogen, progesterone, and combination products exist in various strengths and delivery systems. These products have undergone rigorous review for safety, efficacy, and manufacturing consistency. Both ACOG and the Endocrine Society recommend FDA-approved formulations as the starting point for menopausal hormone therapy.
Compounded HRT becomes clinically relevant in specific circumstances:
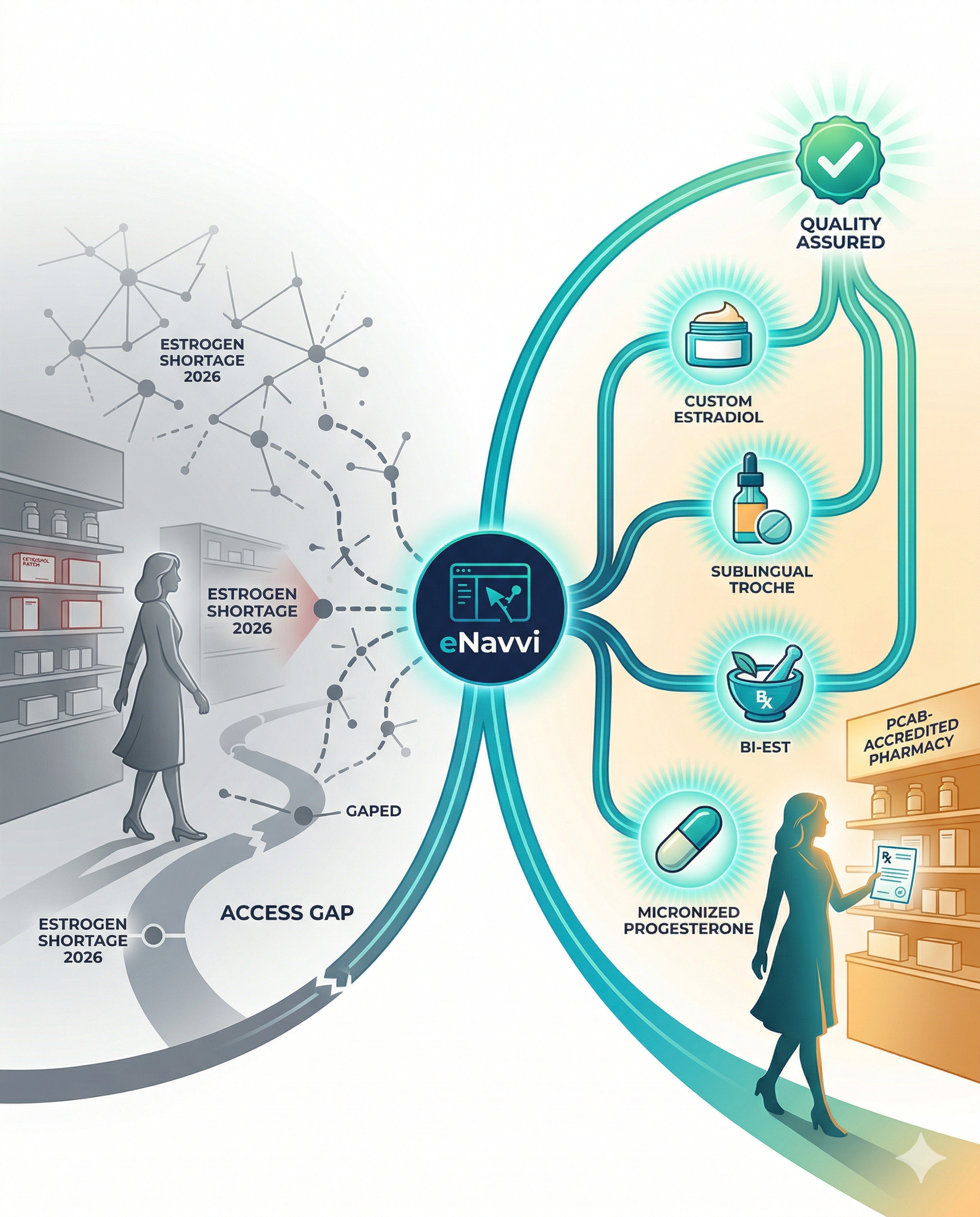
When FDA-approved products are unavailable. The current estrogen patch shortage is a concrete example. When a patient on a stable transdermal estradiol regimen cannot fill her prescription because patches are on backorder, the prescriber faces a clinical decision: switch to an oral formulation (with a different risk profile), switch to a different delivery system the patient hasn’t used before, or consider a compounded transdermal estradiol preparation that delivers the same active ingredient through the same route. The last option maintains therapeutic continuity when the commercial product is temporarily inaccessible.
When the patient needs a non-standard strength or dosage form. FDA-approved products come in fixed doses. Some women require titration to a strength between available options—particularly during the initiation phase or when fine-tuning a regimen for symptom control with minimal side effects. Others need a dosage form that isn’t commercially available: a specific cream concentration, a sublingual troche, or a combination preparation that simplifies a multi-product regimen.
When the patient has allergies or sensitivities to inactive ingredients. Patch adhesives, preservatives, dyes, and fillers in commercial products can cause contact dermatitis or other reactions. Compounding allows the active hormone to be delivered in a vehicle free of the specific allergen—a meaningful clinical consideration for women who react to patch adhesives, which is not uncommon.
When combination therapy is appropriate. Some prescribers prefer to combine estrogen and progesterone—or estrogen with testosterone or DHEA—in a single preparation. While the evidence base for specific multi-hormone compounded combinations is more limited than for individual FDA-approved products, combination compounding is widely used in clinical practice to simplify patient regimens and improve adherence.
The Quality Caveat: Why Pharmacy Selection Is the Clinical Decision
Critics of compounded HRT raise a legitimate concern: quality variability. The National Academies documented potency deviations in compounded hormone preparations as high as 26% from labeled amounts. For hormone therapy—where precise dosing directly affects symptom control, endometrial safety, and cardiovascular risk profile—that variability matters.
But the answer to quality variability is not to deny patients access to compounded hormones when they have a legitimate clinical need. The answer is to send those prescriptions to pharmacies that can document their quality.
PCAB-accredited compounding pharmacies represent approximately 8% of U.S. pharmacies. They undergo on-site inspections, demonstrate compliance with USP ⟨795⟩ and ⟨797⟩ compounding standards, verify ingredient sourcing from FDA-registered suppliers, maintain documented potency testing protocols, and ensure staff competency through ongoing assessment programs. For prescribers who compound HRT, the quality conversation is really a pharmacy selection conversation—and accreditation is the clearest available quality signal.
eNavvi’s network requires PCAB or equivalent accreditation from every pharmacy partner. The strategic alliance with ACHC (announced February 2026) provides member pharmacies with a direct accreditation pathway, including a $1,000 discount on PCAB fees. When a prescriber writes a compounded HRT order through eNavvi, the pharmacy on the other end has met a quality threshold that 92% of U.S. pharmacies have not.
What Prescribers Should Do Right Now
Communicate proactively with patients on estrogen patches. If your patients are on transdermal estradiol, don’t wait for them to call you when the pharmacy can’t fill. Reach out now with a plan: confirm their current prescription status, discuss alternative formulations (gels, creams, oral options), and establish a compounding pharmacy relationship as a backup if their preferred product remains unavailable.
Know the full HRT formulation landscape. The estrogen delivery options go well beyond patches and pills. Transdermal gels and creams (both FDA-approved and compounded), vaginal preparations for genitourinary symptoms, sublingual troches, and injectable estradiol are all available. Compounding pharmacies expand this further with Bi-Est formulations (estriol and estradiol combinations), customizable progesterone preparations, and testosterone or DHEA additions when clinically indicated. Prescribers who understand the full toolkit can pivot quickly when one product becomes unavailable.
Establish your compounding partner before you need one. The worst time to vet a compounding pharmacy is when your patient is mid-shortage and needs a prescription filled tomorrow. Identify a PCAB-accredited pharmacy now—verify their HRT compounding volume, confirm potency testing protocols, and set up your prescribing workflow. Digital platforms like eNavvi make this particularly efficient: browse HRT formulation templates, compare pricing across pharmacies, and send electronic prescriptions without phone calls or faxes.
Counsel patients on the difference between compounding and unregulated products. Shortages drive patients to search for alternatives on their own—including online hormone products, over-the-counter "bioidentical" creams, and unverified supplements. Prescribers have an obligation to explain that physician-prescribed, pharmacy-compounded HRT from an accredited pharmacy is fundamentally different from self-directed purchases of unregulated products. The compounding pathway preserves clinical oversight. The internet pathway does not.
Give Your Patients HRT Options—Even During Shortages
Browse eNavvi’s compounded HRT templates—Bi-Est creams, micronized progesterone, testosterone, and DHEA—with transparent cash pricing across PCAB-accredited pharmacies. Free for prescribers. Your patients shouldn’t have to wait for a supply chain to catch up.
Frequently Asked Questions
Q: Why are estrogen patches in short supply in 2026?
A: The estrogen transdermal patch shortage is driven by a combination of surging demand (HRT prescriptions for women are up 86% since 2021), manufacturer consolidation (only three companies produce the majority of U.S. estradiol patches after Bayer discontinued Climara in 2023), the FDA’s removal of boxed warnings on HRT products (which accelerated prescribing), and tariff pressures on overseas pharmaceutical manufacturing. The shortage is affecting major pharmacy chains nationally.
Q: Is compounded estrogen safe?
A: Compounded estrogen preparations use the same bioidentical hormones (estradiol, estriol) found in FDA-approved products, but they are custom-prepared by compounding pharmacies and are not FDA-approved. Quality can vary between pharmacies, which is why pharmacy selection matters. PCAB-accredited compounding pharmacies are held to rigorous standards for potency testing, ingredient sourcing, and compounding consistency. Prescribers should use compounded HRT when FDA-approved products are unavailable or clinically insufficient, and should always route orders to accredited pharmacies.
Q: What alternatives exist if my patient can’t get estradiol patches?
A: Alternatives to estradiol patches include FDA-approved transdermal gels and sprays, oral estradiol (with a different clot-risk profile), vaginal estrogen preparations for genitourinary symptoms, and compounded transdermal estradiol creams from PCAB-accredited pharmacies. Compounded options offer the advantage of customizable dosing and the same transdermal delivery route as patches, maintaining therapeutic continuity for patients who prefer to avoid oral formulations. Prescribers can compare options and pricing on platforms like eNavvi.